Abstract:
The migratory properties of leukemic cells are commonly associated with their pathological potential and can significantly affect the disease progression. While the research in immunopathology mostly employed powerful indirect methods such as flow cytometry, these cells were rarely observed directly using live imaging microscopy. This is especially true for the malignant cells of the B-cell lineage, such as those originating from chronic lymphocytic leukemia (CLL) and mantle cell lymphoma (MCL). In this study, we employed open-source image analysis tools to automatically and quantitatively describe the amoeboid migration of four B-cell leukemic and lymphoma cell lines and primary CLL cells. To avoid the effect of the shear stress of the medium on these usually non-adherent cells, we have confined the cells using a modified under-agarose assay. Surprisingly, the behavior of tested cell lines differed substantially in terms of basal motility or response to chemokines and VCAM1 stimulation. Since casein kinase 1 (CK1) was reported as a regulator of B-cell migration and a promoter of CLL, we looked at the effects of CK1 inhibition in more detail. Migration analysis revealed that CK1 inhibition induced rapid negative effects on the migratory polarity of these cells, which was quantitatively and morphologically distinct from the effect of ROCK inhibition. We have set up an assay that visualizes endocytic vesicles in the uropod and facilitates morphological analysis. This assay hints that the effect of CK1 inhibition might be connected to defects in polarized intracellular transport. In summary, 1) we introduce and validate a pipeline for the imaging and quantitative assessment of the amoeboid migration of CLL/MCL cells, 2) we provide evidence that the assay is sensitive enough to mechanistically study migration defects identified by the transwell assay, and 3) we describe the polarity defects induced by inhibition or deletion of CK1ε.
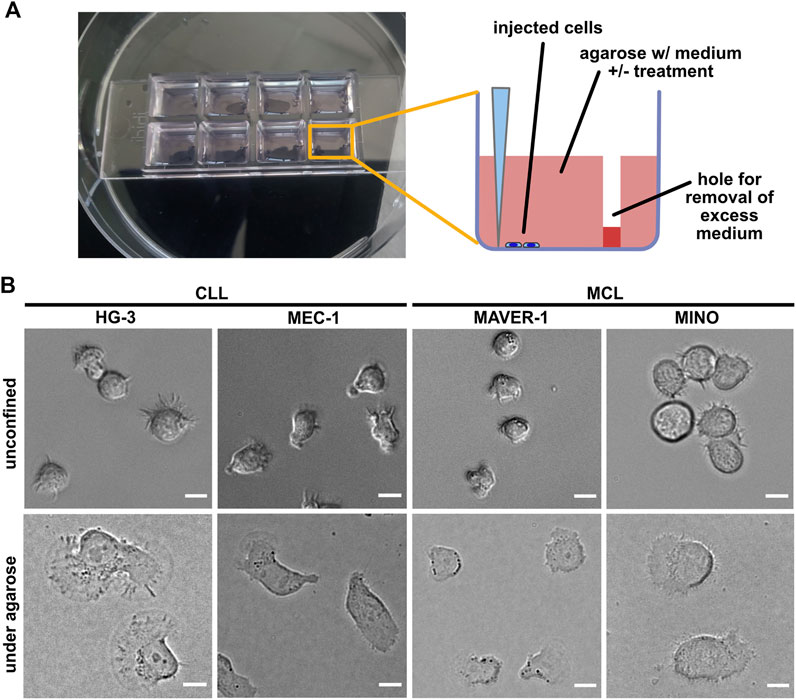
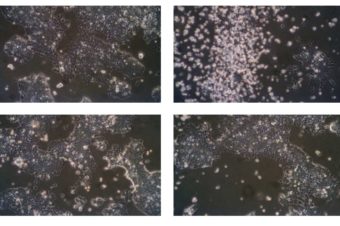
FIGURE 1. Experimental system for analysis of ameboid migration of leukemic cell lines. (A) Scheme of the under-agarose assay experimental system used for migration analysis. The cells are injected under the gel using a 10-μl pipette at the side of the well opposite to hole punched in advance, which serves to collect excess medium. The collected medium needs to be removed before imaging to achieve efficient confinement. Inhibitors or chemokines are added to the gelation mixture before the gel is poured into the wells. (B) Representative images of cell lines used in this study. Upper line: cell phenotypes in the normal 2D culture. Lower line: cell phenotypes in the under-agarose assay. Scale bar: 10 μm. Representative videos of cell line behavior under confinement are located in Supplementary Movies S1–S4.
Authors:
Štěpán Čada 1, Olga Vondálová Blanářová 1, Kristína Gömoryová 1, Antónia Mikulová 1, Petra Bačovská 1, Nikodém Zezula 1, Alka Kumari Jadaun 1, Pavlína Janovská 1, Hana Plešingerová 1,2, Vítězslav Bryja 1,3
- 1 Department of Experimental Biology, Faculty of Science, Masaryk University, Brno, Czechia.
- 2 Department of Internal Medicine-Hematology and Oncology, University Hospital Brno, Brno, Czechia.
- 3 Department of Cytokinetics, Institute of Biophysics, Academy of Sciences of the Czech Republic, Brno, Czechia